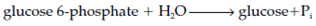Glycogen degradation:
Glycogen degradation needs two enzymes; glycogen-debranching enzyme and glycogen phosphorylase.
Glycogen phosphorylase frequently called simply phosphorylase degrades glycogen through breaking α1-4 glycosidic bonds to release glucose units one at a time from the nonreducing end of the glycogen molecule (the end with a free 4' OH group) as glucose 1-phosphate. The other substrate needs is inorganic phosphate (Pi). The reaction is an instance of phosphorolysis, which is breakage of a covalent bond through the addition of a phosphate group. The (reversible) reaction is as given below:
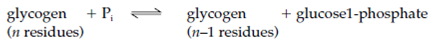
Moreover, glycogen phosphorylase can erased only those glucose residues which are more than five residues from a branchpoint. Glycogen-debranching enzyme erased the α1-6 branches and so permits phosphorylase to continue degrading the glycogen molecule. The glucose 1-phosphate formed is converted to glucose 6-phosphate through the enzyme phosphoglucomutase:
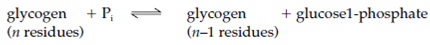
The fate of the glucose 6-phosphate depends on the tissue. Liver holds the enzyme glucose 6-phosphatase that converts the glucose 6-phosphate to glucose, that then diffuses out into the bloodstream and so maintains the blood glucose concentration:

In During glycogen degradation in muscle, the major goal is to produce energy rapidly and so the glucose 6-phosphate is metabolized immediately through glycolysis. This tissue does not hold glucose 6-phosphatase.