Counting prokaryotes
In microbiology the number of cells per milliliter of sample is often important. We can be fairly sure of finding most species of prokaryote in a sample give that sample is large enough and was taken from a habitat which allows growth of that organism. It is likely which we might search Salmonella on eggs but the important question we require to answer is the eggs safe to eat? To be safe, there must be less than the intoxicating dose of Salmonella in an amount of egg which is likely to be consumed raw. The number of cells by to cause Salmonella food poisoning is around 40 so we could reasonably expect safe eggs to carry less than example for 40 cells per dozen eggs. There is a limit exist in organizational standards for most foods, so a good estimation of bacterial numbers is crucial to the food organization. In medicine, the presence of only one or two Staphylococcus aureus per 10 cm2 of human skin could be considered quite normal but 104 cells per mm2 might reveal the underlying cause of a serious skin condition. In environmental microbiology the relative numbers of organisms per ml of a sample of river water might specify the dominant species.
Numbers of prokaryotes in a sample can be expressed in two ways that are: the total count or the viable count. The former estimates the number of alive, cells or dead the latter only those capable of growing under the conditions tested. Total counts are made through diluting the sample in a known amount of buffer and then counting the number of cells in each well of a hemocytometer. The hemocytometer is a specialized microscope covers and slide slip in that a grid of known size is displayed while viewing under the microscope. The count of cells per grid can then be multiplied up to reveal the number of cells per ml in the original sample. Flow cytometry is a technique same in concept in which the number of particles in a small sample is electronically counted through passing a laser shining across a capillary approximately one cell broad.
More recently these techniques have been complemented through quantitative PCR. This method allows the counting of the number of copies of individual genes. For example if the copy number per ml of the 16S rRNA gene is estimated this can give an idea of the bacterial numbers. Otherwise there are drawbacks to this method we can simultaneously estimate the relative numbers of several different prokaryotes and eukaryotes through carrying out parallel experiments on the similar sample using specific primer sets. Whether cells are alive or dormant can be related to the presence of ATP. Therefore, an estimate can be made of the overall activity of a sample through measuring ATP. Similarly, only live cells can transcribe DNA, so an estimation of viability can be made through measuring mRNA concentrations from ribosomal genes using reverse-transcription. This presupposes which only active cells would accumulate these transcripts.
The most generally used and informative technique for enumeration of prokaryotes is the viable count. Classically, viable counts are made by serial dilution. This agar plate-based method gives a result in colony forming units (cfu) ml-1. This is not equivalent to the true viable count as the numbers only reflect those species which are capable of forming visible colonies under the conditions of medium and incubation chosen for the experiment. The number of cells in the original sample is estimated through back-calculating the number of dilutions made from the plate which has the highest number of easily discernible colonies. Normally this figure is less than 200 colonies per plate but varies according to colony size. The equivalent method for prokaryotes not capable of growth on agar is the most probable number methods in that the pattern of growth in replicates of liquid cultures at several dilutions is used to reduce the number in the real sample.
One of the most accurate ways of counting any microscopic particle is through use of FACS. Fluorescent dyes can be obtained which differentially stain dead and living cells. The illustration is introduced into a narrow capillary of only one
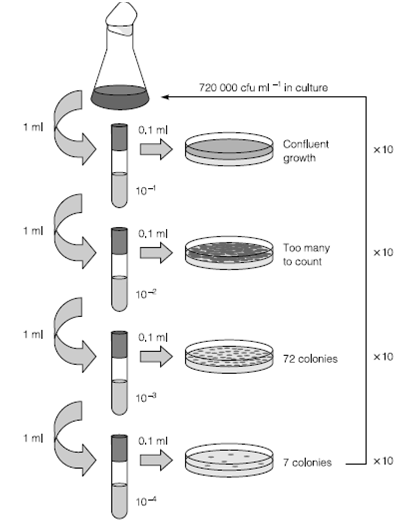
Figure 2: Diagram showing serial dilutions for the estimation of viable count. Test tubes contain 9 ml before addition of bacterial culture.
cell width in diameter and passes through a laser. The laser excites the fluorescent dye and the excitation passes to a detector. The detector is linked to a gate just downstream of the laser that will switch to move the cell into a receptacle. Nonfluorescent cells do not cause the gate to open and pass into a second vessel. The number of times the excitation gate opens can be counted and related to the flow rate previous the laser to give an specific number of cells in the sample.