Reference no: EM132812829
Question 1. Define the following terms
I; Half life (t1/2): .....................................................................
II; Radioactivity: .....................................................................
III; Isotopes: ..........................................................................
IV; Isobars: ..............................................................................
Question 2. (a) State two differences between nuclear and chemical reactions
Table 1
| Nuclear reaction |
Chemical reaction |
1........................................
2........................................ |
1.................................................
2................................................. |
(b) The reaction scheme below shows a radioactive decay series starting from 21483Bi Study the scheme and answer the questions that follow.
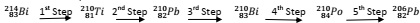
(i) Identify the particles emitted at; 3rd step; ...................
5th step; ...................
(ii) Write the nuclear equation for the reaction which took place in the 1st step.
........................................
3). Table 2 shows the percentage of radioactive isotopes of bismuth remaining after decay at different times
Table 2
| Time (Minutes) |
0 |
6 |
12 |
22 |
38 |
62 |
100 |
| Percentage of Bismuth |
100 |
81 |
65 |
46 |
29 |
12 |
3 |
(i) On the grid provided, plot a graph of percentage (vertical axis) against time

(ii) Using the graph, determine;
I; The half life of Bismuth
..................................................
II; The original mass of bismuth, given that the mass remaining after 70 minutes was 0.16 g
...................................................
(4) Table 3 gives the rate of decay for a radioactive isotope A (where A is not the actual symbol for the element)
Table 3
| Number of days |
Mass (g) |
| 0 |
400 |
| 240 |
50 |
Calculate the half life (t1/2) of element A
(5) One hundred grams (100 g) of a radioactive 23391Pa was reduced to 6.25 g after 108 days. Determine the half life (t1/2) of 23391Pa.
(6) Q grams of a radioactive isotope decayed to10 g in 80 days. The half life of the isotope is 20 days. Calculate the initial mass Q of the radioactive isotope.
(7) After 45 days, 1/8 of the mass of a radioactive isotope remained. Determine its half life (t1/2).
(8) Determine the values of T1 and T2 in the nuclear equation below.
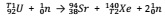
T1; ...................................
T2; ...................................
(9) Explain the source of electrons in a radioactive process.
(10) State dangers associated with the exposure of human beings to radioisotopes
(11) State one application of radioactive isotopes in;
I; Medicine...............................................................
II; Agriculture.........................................................
III; Industry............................................................
IV; Nuclear power stations................................
(12) State two differences between nuclear fusion and fission;
| Fusion |
Fission |
1................................................
2................................................ |
1.......................................
2....................................... |
13). Complete diagram 1 to show how particles are distinguished. Label the diagram clearly.

diagram 1
14). Explain why it is not safe to store radioactive substances in containers made from aluminum sheets.
15) A radioisotope B2 decays by emitting three alpha (α) particles and two beta (β) to from 21483Bi.
Determine the atomic and mass numbers of B2?
Atomic number; ..............................
Mass number; .................................
16) Describe how radioactive solid wastes should be disposed
Attachment:- RADIOACTIVITY.rar