Reference no: EM133353138
Chemical Instrumental Analysis
Question #1
In an experiment, we created a set of 8 concentrations of 2 dyes (one red and one yellow), then used a spectrophotometer to calculate the absorbance of the dyes with respect to both concentration and wavelength. Our ultimate goal was to calculate the concentrations of each dye in a solution containing a mixture of both of them. The concentrations of the red and yellow dyes in the unknown. λ1 and λ2 are two different wavelengths, selected to have the greatest differences in absorbance between the two dyes.
The absorbance A1 at λ1 (423.8 nm) is 1.599. Similarly, the absorbance A2 at λ2 (501.3 nm) is 0.771. Note that these wavelengths correlate with peaks of the individual dyes' absorption spectra. The molar extinction coefficients are measured in the experiment to be:
0.0160 L mol-1 cm-1 at 423.8nm for the red dye.
0.0341 L mol-1 cm-1 at 423.8nm for the yellow dye.
0.0351 L mol-1 cm-1at 501.3nm for the red dye.
0.0023 L mol-1 cm-1at 501.3nm for the yellow dye.
Find the concentrations of the red and yellow dyes in the unknown.
Question #2
White light is sent through an interface of a 100% (w/v) glycerol solution (??1=1.474) and a 20% (w/v) sucrose solution (??2=1.364) at an angle of 340.
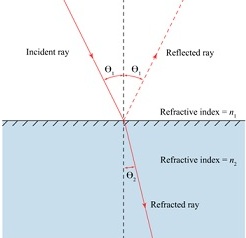
If Θ1=34°, determine the angle of Θ2 in degrees.
Question #3
This is the FTIR spectrum of HCTZ a drug in the category of thiazide and thiazide-like diuretics. Label the important features in the spectra.
This is the structure of HCTZ:
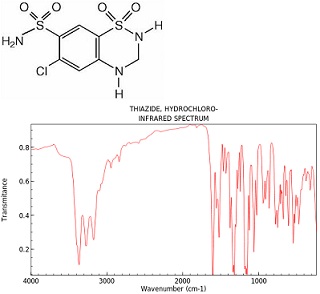
The area of the peak around 1200 cm-1 has a value of 20000 units. This is the FTIR of the drug HCTZ in pill form.
An FTIR for the urine output for a patient is the same with the peak area at 1200 cm-1is 16000 units.
What percentage of the drug is unmetabolized in the patient.
The yellow color emitted by sodium in a flame test arises from two strong emission lines, one centered at 588.995 0 nm and the other at 589.592 4 nm. These are known as the sodium D-lines and arise from electrons falling from the 3p orbital to the 3s orbital. The line at 589.592 4 nm has a natural linewidth of 0.33 × 10-3 cm-1.
What is the natural linewidth in nm?
Question #4
Diagram, label, and describe how a PMT works.
Question #5
The wavelength of maximum exitance from a blackbody at a given temperature can be estimated from the Wien displacement law:
λmaxT = 2.90 x 103 μm.K
Calculate the temperature (in K) required for λmax to correspond to the following:
The wavelength of maximum absorbance of an aqueous solution of potassium permanganate (525 nm)
Question #6
A schematic of a double beam spectrophotometer is shown.
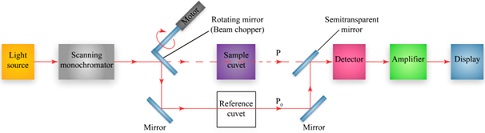
Identify the part of the spectrophotometer with its description.
____________________________ houses the reagent blank
____________________________ passes the beam to the detector
____________________________ holds the sample with a defined pathlength
____________________________ increases the detector signal for ease of display and quantitation
____________________________ mirror that rotates to direct light to either the sample of the reference cuvet
____________________________ provides electromagnetic radiation in the ultraviolet (UV), visible, or infrared (IR) region of the spectrum
____________________________ typically a photomultiplier tube that generates an electrical current proportional to the photon flux
____________________________ wavelength selector that passes a narrow band of wavelengths to the sample by scanning through a user defined range of selected wavelengths.