Reference no: EM131083655
The acid-base indicator Hln undergoes the following reaction in dilute aqueous solution:
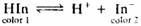
The following absorbance data were obtained for a 5.00 X 10-4 M solution of HIn in 0.1 M NaOH and 0.1 M HCI. Measurements were made at wavelengths of 485 nm and 625 nm with 1.00·cm cells.
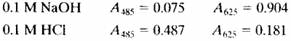
In the NaOH solution, essentially all of the indicator is present as In; in the acidic solution, it is essentially all in the form of HIn.
(a) Calculate molar absorptivities for In- and HIn at 485 and 625 nm.
(b) Calculate the acid dissociation constant for the indicator if a pH 5.00 buffer containing a small amount of the indicator exhibits an absorbance of 0.567 at .+85nm and 0.395 at 625 nm (1.00-cm cells).
(c) What is the pH of a solution containing a small amount of the indicator that exhibits an absorbance of 0.492 at 485 nm and 0.2'+5 at 635 nm (1.00-cm cells)?
(d) A 25.00-mL aliquot of a solution of purified weak organic acid HX required exactly 24.20mL of a standard solution of a strong base to reach a phenolphthalein end point. When exactly 12.10 mL of the base was added to a second 25.00-mL aliquot of the acid, which contained a small amount of the indicator under consideration, the absorbance was found to be 0.333 at 485 nm and 0.655 at 625 nm (1.00-cm cells). Calculate the pH of the solution and Ka for the weak acid.
(e) What would be the absorbance o[ a solution at 485 and 625 nm (1.50-cm cells) that was 2.00 x 10-4 M in the indicator and was buffered to a pH of 6.000?
|
Calculate the company debt-to-equity ratio
: Boulder Mountain Ski Company has total assets of $474,300,000 and a debt ratio of 0.30. Calculate the company's debt-to-equity ratio.
|
|
Question regarding the level of inventory
: Soaring Eagles Corp. has total current assets of $11,372,000, current liabilities of $5,572,000 and a quick ratio of 0.85. What is its level of inventory?
|
|
Write a primary source analysis paper about muslim empires
: Write a three to four page primary source analysis paper about Muslim Empires. Please be plagarism free, paper will be submitted through plagarism checker.
|
|
What are the appropriate uses of a budget
: Why is budgeting important? What are the appropriate uses of a budget? How can budgets be mismanaged or misunderstood? What are some criteria you would use to choose between a fixed and a flexible budget? Explain your answers.
|
|
Calculate the acid dissociation constant for the indicator
: Calculate molar absorptivities for In- and HIn at 485 and 625 nm.
|
|
Firm accounts receivables turnover
: Blue Air Inc., has net sales of $756,000 and accounts receivables of $160,000. What are the firm's accounts receivables turnover?
|
|
What would you tell your cousin about this disease
: Imagine your 19 year old cousin has recently gone through many changes in his life and you suspect he is suffering from major depression. What would you tell your cousin about this disease? How would you help him find the best treatment possible? ..
|
|
Compare structure and character of political institutions
: Compare and contrast the structure and character of political institutions in the Greek city-states with political institutions in other early western civilizations we have explored to date in class.
|
|
Regulations in the financial sector
: What regulations in the financial sector are likely to grow in the future
|