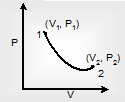
Thermodynamic process is the path given by the confined gas in going from one thermodynamic phase to another state.
This phenomena is represented by 1 → 2
(i) Cyclic process: When a system returns back to its initial state after passing through different phase, the phenomena is said to be a cyclic process. In this process since ΔU = 0.
From first law of thermodynamics, ΔQ = W
Thus in a cyclic phenomena, the heat absorbed by the system is equal to the net work done by the object. Work done is the area of the cycle on the PV diagram.
|
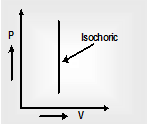
(ii) Isochoric process: a thermodynamic process, in which the volume of gas always constant is called isochoric process. In this process, the work done by the body is zero.
Therefore from first law of thermodynamics, ΔQ = ΔU as the net work done Wisochoric = 0 Thus in an isochoric process, the total heat energy absorbed by the system is used to change its internal energy.
|
|
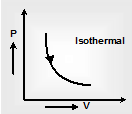
(iii) Isothermal process: In an isothermal process, the temperature of body always constant throughout the process. Since the internal energy of a system depends only on its temperature, therefore there is no modification in internal energy of a perfect gas (ideal gas) in an isothermal process.
i.e. ΔU = 0 since PV = nRT so, for T = constant PV = constant This represent a rectangular hyperbola as shown. Net work done = 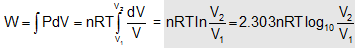 |
|
(iv) Adiabatic process: In an adiabatic phenomena, the body neither absorbs heat nor does it reject energy, i.e. in this phenomena there is no heat exchange (ΔQ = 0).
Therefore from first law of thermodynamics, W = -ΔU
(v) Isobaric process: In isobaric process pressure of the gas is constant throughout the process. Hence, V/T is constant in isobaric process.
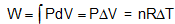
|
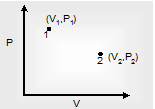
(vi) Free Expansion process: In free expansion of gas, the gases expand in vacuum.
Therefore, work done Δw = 0
In free expansion if state changes from 1→ 2 only the initial and final states are known intermediate states are not given. Therefore a path cannot be given as in the figure from 1 to 2.
|
|
Email based Physics assignment help - homework help at Expertsmind
Are you searching physics expert for help with Thermodynamic process questions? Thermodynamic process topic is not easier to learn without external help? We at www.expertsmind.com offer finest service of Physics assignment help and physics homework help. Live tutors are available for 24x7 hours helping students in their Thermodynamic process related problems. We provide step by step Thermodynamic process question's answers with 100% plagiarism free content. We prepare quality content and notes for Thermodynamic process topic under physics theory and study material. These are avail for subscribed users and they can get advantages anytime.
Why Expertsmind for assignment help
- Higher degree holder and experienced experts network
- Punctuality and responsibility of work
- Quality solution with 100% plagiarism free answers
- Time on Delivery
- Privacy of information and details
- Excellence in solving physics queries in excels and word format.
- Best tutoring assistance 24x7 hours