Organometallic compounds
These are the compounds in which a metal atom or a metalloid (Ge, Sb) or a non-metal atom such as B, P, Si, etc, (less electronegative than C) is directly linked to a carbon atom of a hydrocarbon radical or molecule. Organometallic the compounds have at least one.
(1) Metal - Carbon bond, (2) Metalloid - Carbon bond, (3) Non metal - Carbon bond.
Example :
Compounds : C2H5MgBr, (C2H5)2Zn, C6H5Ti(OC3H7)3, (CH3)4Si
Organometallic bond: Mg-C, Zn-C, Ti-C, Si-C
Classification of organometallic compounds : Organometallics have been classified as :
(1) σ -bonded organometallic compounds : Compounds such as RMgX, R2Zn, R3Pb, R3Al, R4Sn etc, contains M-C σ - bond and are called σ - bonded organometallic compound.
(2) Π - bonded organometallic compounds : The transition metals binds to unsaturated hydrocarbons and their derivatives using their d-orbitals. The metal atom hear is bonded to ligands in such a way that donations of electrons and back acceptance by the ligand is feassible. These are called Π - orbitals of the ligand. These are called Π - complexes.
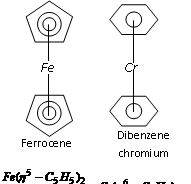
Examples : (i) Π - cyclopentadienyl - iron complex
Ferrocene 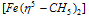 , Bis (cyclopentadienyl) iron (II)
, Bis (cyclopentadienyl) iron (II)
It is the Π bonded sandwich compound. Number of the carbon atoms bonded to metal ion is indicated by the superscript on eta 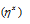 that is
that is 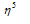 in this complex.
in this complex.
(ii) Dibenzene chromium ( Π - complex)
It is also a Π - bonded sandwitch compound. Its formula can be given as 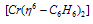
(iii) Alkene complex ( Π - complex)
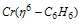
Zeise's salt K 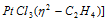 ; Potassium trichloroethylene platinate (IV).
; Potassium trichloroethylene platinate (IV).
It is a Π - bonded complex. μ2 indicates that two carbons of ethylene are bonded to metals.
(3) Complexes containing both s- and p-bonding characteristics : The metal carbonyls, compounds formed among metal and carbon monoxide belong to this class. The metal carbonyls have been included in the organometallics.
(i) Mononuclear carbonyls : Contain one metallic atom per molecule. The example of which is given as follows Ni(CO)4, Fe(CO)5, Cr(CO)6
(ii) Polynuclear carbonyls : These contain two or more metallic atoms per molecule. The example of which is given as follows
Mn2(CO)10, Fe(CO)9, Fe(CO)12
Applications of organometallics
(1) Grignard reagent (RMgX) has been extensively used for synthesis of various organic compounds.
(2) Wilkinson's catalyst [(PH3P)3RhCl i.e. tris (triphenylphosphine) chlororhodium (I) is used as a homogeneous catalyst for the hydrogenation of alkenes.
(3) Zeigler Natta catalyst (composed of a transition metal salt, generally TiCl4 and trialkyl aluminium) are used as heterogeneous catalysts in the polymerisation of alkenes.
Email based Chemistry assignment help - homework help at Expertsmind
Are you searching chemistry expert for help with Organometallic compounds questions? Organometallic compounds topic is not easier to learn without external help? We at www.expertsmind.com offer finest service of Chemistry assignment help and chemistry homework help. Live tutors are available for 24x7 hours helping students in their Organometallic compounds related problems. We provide step by step Organometallic compounds question's answers with 100% plagiarism free content. We prepare quality content and notes for Organometallic compounds topic under chemistry theory and study material. These are avail for subscribed users and they can get advantages anytime.
Why Expertsmind for assignment help
- Higher degree holder and experienced experts network
- Punctuality and responsibility of work
- Quality solution with 100% plagiarism free answers
- Time on Delivery
- Privacy of information and details
- Excellence in solving chemistry queries in excels and word format.
- Best tutoring assistance 24x7 hours