Factors affecting the value of ionisation energy
(i) Size of atom : With an increase in the atomic size, ionisation potential is reduced, as the distance of the outermost electron from nucleus increases and hence the force of attraction decreases.
(ii) The charge on the nucleus : With an increase in the nuclear charge, there is an increase in the force of attraction of the nucleus for electrons making the removal of the electrons more difficult. Therefore an increase in nuclear charge increases the ionisation potential.
(iii) The shielding or screening effect of inner shells : The valence electrons in a multi-electron atom are pulled by the nucleus but are repelled by the electrons of the inner shells. The valence electrons, so, do not experience the total pull of the nucleus. Instead the whole pull of the nucleus is reduced by the electrons in inner shells. The effect of reducing the force of attraction of nucleus by the inner shells is called as screening effect. This effect is exhibited maximum by s2p6 (the most stable) shell. Hence, the ionisation energy of K is much less than Cu, though, both have one electron in their fourth shell.
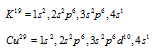
The ionisation energy of K is 4.33 eV while of the Cu is 7.72 eV. This is because of a large screening effect of s2p6 penultimate orbit in K while s2p6d10 penultimate orbit in Cu which exhibit little screening effect.
(iv) Type of electrons involved: Ionisation energy also depends upon the type that is s, p, d or f, electrons which are to be removed, s -electrons are closer to the nucleus and are more firmly held as compared to p, d or f electrons. Hence, ionisation energy decreases in the order of s > p > d > f orbitals.
(v) Completely filled or half filled sub-shells : According to Hund's rule, fully filled or half filled orbitals are more stable. As a result, it is comparatively difficult to remove the electrons from these shells. The ionisation energy of Be (9.3 eV) is more than B (8.3 eV) because Be has 2s2 configuration of the outermost orbit which is fully filled. Similarly, nitrogen (14.6 eV) has more ionisation energy than oxygen (13.6 eV) because nitrogen has outermost shell configuration as 2s2p3 in which p shell is half filled and is more stable. Similarly, ionisation energy of Mg is more than Al and that of P is more than S.
Email based Chemistry assignment help - homework help at Expertsmind
Are you searching chemistry expert for help with Factors affecting the value of ionisation energy questions? Factors affecting the value of ionisation energy topic is not easier to learn without external help? We at www.expertsmind.com offer finest service of Chemistry assignment help and chemistry homework help. Live tutors are available for 24x7 hours helping students in their Factors affecting the value of ionisation energy related problems. We provide step by step Factors affecting the value of ionisation energy question's answers with 100% plagiarism free content. We prepare quality content and notes for Factors affecting the value of ionisation energy topic under chemistry theory and study material. These are avail for subscribed users and they can get advantages anytime.
Why Expertsmind for assignment help
- Higher degree holder and experienced experts network
- Punctuality and responsibility of work
- Quality solution with 100% plagiarism free answers
- Time on Delivery
- Privacy of information and details
- Excellence in solving chemistry queries in excels and word format.
- Best tutoring assistance 24x7 hours