Chemical properties
(i) Addition reaction: The carbonyl group is polar as oxygen is more electronegative than carbon,
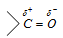
Thus, the positive part of the polar reagent always goes to the carbonyl oxygen and negative part goes to carbonyl carbon.
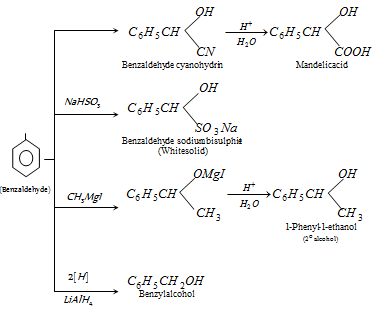
However on reduction with sodium amalgam and water, it gives hydrobenzoin,
(ii) Clemmensen's reduction : With amalgamated zinc and conc. HCl, benzaldehyde is reduced to toluene.
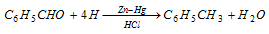
(iii) Schiff's reaction: It restores pink colour to Schiff's reagent (aqueous solution of p-rosaniline hydrochloride decolourised by passing sulphur dioxide).
(iv) Tischenko reaction : On heating benzaldehyde with aluminium alkoxide (ethoxide) and a little of anhydrous AlCl3 or ZnCl2, it undergoes an intermolecular oxidation and reduction (like aliphatic aldehydes) to form acid and alcohol respectively as such and react to produce benzyl benzoate (an ester).
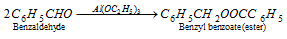
(v) Reactions in which benzaldehyde differs from aliphatic aldehydes
(a) With fehling's solution : No reaction
(b) Action of chlorine : Benzoyl chloride is formed when chlorine is passed through benzaldehyde at its boiling point in absence of halogen carrier. This is because in benzaldehyde there is no α-hydrogen atom present which could be replaced by chlorine.
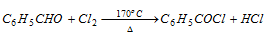
(c) Cannizzaro's reaction : 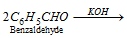
(d) Benzoin Condensation
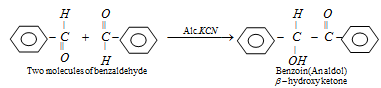 (β-hydroxy ketone)
(β-hydroxy ketone)
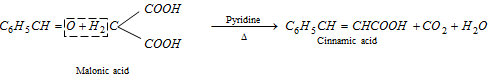
(e) Perkin's reaction
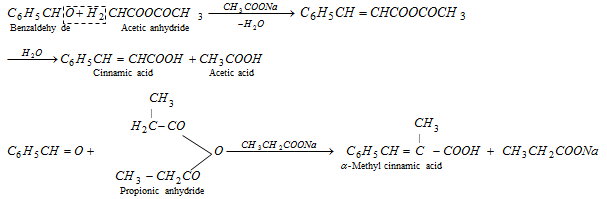
(f) Claisen condensation [Claisen-schmidt reaction]
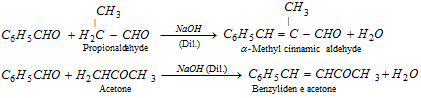
(g) Knoevenagel reaction
(h) Reaction with aniline : Benzaldehyde reacts with aniline and forms Schiff's base
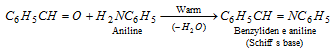
(i) Reformatsky reaction
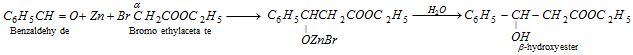
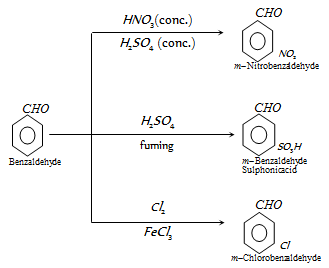
(k) Reaction of benzene ring
Email based Chemistry assignment help - homework help at Expertsmind
Are you searching chemistry expert for help with Chemical Properties of Benzaldehyde questions? Chemical Properties of Benzaldehyde topic is not easier to learn without external help? We at www.expertsmind.com offer finest service of Chemistry assignment help and chemistry homework help. Live tutors are available for 24x7 hours helping students in their Chemical Properties of Benzaldehyde related problems. We provide step by step Chemical Properties of Benzaldehyde question's answers with 100% plagiarism free content. We prepare quality content and notes for Chemical Properties of Benzaldehyde topic under chemistry theory and study material. These are avail for subscribed users and they can get advantages anytime.
Why Expertsmind for assignment help
- Higher degree holder and experienced experts network
- Punctuality and responsibility of work
- Quality solution with 100% plagiarism free answers
- Time on Delivery
- Privacy of information and details
- Excellence in solving chemistry queries in excels and word format.
- Best tutoring assistance 24x7 hours