Bronsted-Lowry concept : In accordance to this concept,
An acid is defined as a substance which has the tendency to give a proton (H+) and a base is defined as a substance which has a tendency to accept a proton. Or we can say that, an acid is a proton donor whereas a base is a proton acceptor.
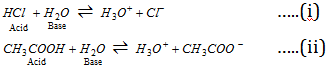
(i) HCl and CH3COOH are acids because they donate a proton to H2O.(ii) NH3 and CO32- are bases because they accept a proton from water.
In reaction (i), in reverse process of it, H3O+ can provide a proton and thus is an acid while Cl- can accept the proton and hence is a base. Therefore there are two acid-base pairs in reaction (i). These are HCl - Cl- and H3O+- H2O. These acid-base pairs are called conjugate acid-base pairs.
Conjugate acid ? Conjugate base + H+
Conjugate base of a strong acid is a weak base and vice a versa. Weak acid has a strong conjugate base and vice a versa.
Email based Chemistry assignment help - homework help at Expertsmind
Are you searching chemistry expert for help with Bronsted–Lowry concept questions? Bronsted–Lowry concept topic is not easier to learn without external help? We at www.expertsmind.com offer finest service of Chemistry assignment help and chemistry homework help. Live tutors are available for 24x7 hours helping students in their Bronsted–Lowry concept related problems. We provide step by step Bronsted–Lowry concept question's answers with 100% plagiarism free content. We prepare quality content and notes for Bronsted–Lowry concept topic under chemistry theory and study material. These are avail for subscribed users and they can get advantages anytime.
Why Expertsmind for assignment help
- Higher degree holder and experienced experts network
- Punctuality and responsibility of work
- Quality solution with 100% plagiarism free answers
- Time on Delivery
- Privacy of information and details
- Excellence in solving chemistry queries in excels and word format.
- Best tutoring assistance 24x7 hours