Benzaldehyde, C6H5CHO
Benzaldehyde is the simplest aromatic aldehyde. It happens in bitter almonds in the form of its glucoside, amygdalin (C20H27O11N). When amygdalin is boiled with dilute acids, it hydrolyses goes into benzaldehyde, HCN nad glucose.
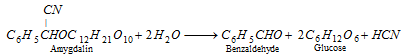
Benzaldehyde is also defined as oil of bitter almonds.
(1) Method of preparation
(i) Laboratory method : It is conveniently prepared by boiling benzyl chloride with copper nitrate or lead nitrate solution in a current of carbon dioxide.
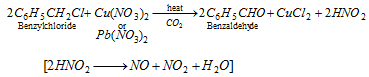
(ii) Rosenmund reaction :
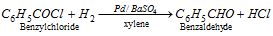
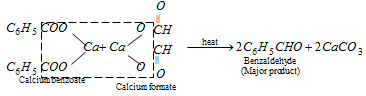
(iii) By dry distillation of a mixture of calcium benzoate and calcium formate
(iv) By oxidation of benzyl alcohol : This involves the treatment of benzyl alcohol with dil. HNO3 or acidic potassium dichromate or chromic anhydride in acetic anhydride or with copper catalyst at 350o C.
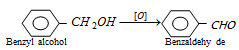
This process is used for commercial production of benzaldehyde.
(v) By oxidation of Toluene
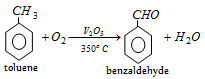
Commercially the oxidation of toluene is done with air and diluted with nitrogen (to prevent complete oxidation) at 500o C in the presence of oxides of Mn, Mo or Zr as catalyst.
(vi) Etard's reaction : 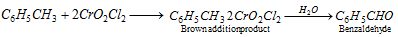
(vii) Gattermann-koch aldehyde synthesis : Benzene is converted into benzaldehyde by passing a mixture of carbon monoxide and HCl gas under high pressure into the ether solution of benzene in presence of anhydrous aluminium chloride and cuprous chloride.
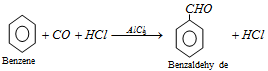
(viii) Gattermann reaction
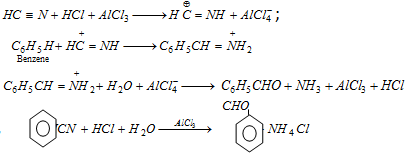
(ix) Stephen's reaction : Benzaldehyde is obtained by partial reduction of phenyl cyanide with stannous chloride and passing dry HCl gas in ether solution followed by hydrolysis of the aldimine stannic chloride with water.
(x) By ozonolysis of styrene
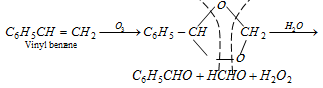
(xi) Grignard reaction
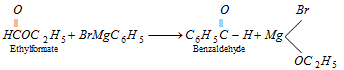
Other reagents like carbon monoxide or HCN can also be used in place of ethyl formate.
Email based Chemistry assignment help - homework help at Expertsmind
Are you searching chemistry expert for help with Benzaldehyde questions? Benzaldehyde topic is not easier to learn without external help? We at www.expertsmind.com offer finest service of Chemistry assignment help and chemistry homework help. Live tutors are available for 24x7 hours helping students in their Benzaldehyde related problems. We provide step by step Benzaldehyde question's answers with 100% plagiarism free content. We prepare quality content and notes for Benzaldehyde topic under chemistry theory and study material. These are avail for subscribed users and they can get advantages anytime.
Why Expertsmind for assignment help
- Higher degree holder and experienced experts network
- Punctuality and responsibility of work
- Quality solution with 100% plagiarism free answers
- Time on Delivery
- Privacy of information and details
- Excellence in solving chemistry queries in excels and word format.
- Best tutoring assistance 24x7 hours